|
|||||
|
|||||||
|
    An initiative of :Stichting Food-Info
|
| Food-Info.net> Topics > Food components > Food colours > Natural food colours > Anthocyanins and anthocyanidins Anthocyanins and anthocyanidinsAnthocyanins are a very large group of red-blue plant pigments. Anthocyanins occur in all higher plants, mostly in flowers and fruits but also in leaves, stems, and roots. In these parts they are found predominantly in outer cell layers. The amounts are relatively large: one kilogram of blackberry for example contains approximately 1.15 gram, and red and black legumes can contain 20 mg per gram. The colour of anthocyanins depends on the structure, but also on the acidity of the fruit. Many antocyanins are red at acidic conditions and turn blue at less acid conditions. Chemically anthocyanins are subdivided into the sugar-free anthocyanidine aglycons and the anthocyanin glycosides. They are used as food additive with E number E163.  Fig. 1: Blueberries, a rich source of anthocyanins (Source) StructureOver 500 different anthocyanins have been isolated from plants. They are all based on a single basic core structure, the flavyllium ion (See figure 2). 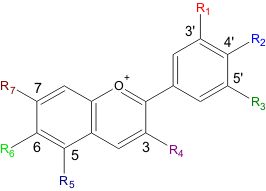 Fig. 2 : The flavylium ion, the basic structure of anthocyanins. (Source) As shown in figure 2 there are 7 different side groups on the flavylium ion. These side groups can be a hydrogen atom, a hydroxide or a methoxy-group. The most frequent combination of side groups and their names are shown in Table 1. Table 1 : Main groups of anthocyanidins. R1-R7 are the side groups as shown in figure 2
When anthocyanidins are coupled to sugars, anthocyanins are formed. As sugars can be coupled at different places and many different sugars are present in plants, it is clear that a very large range of anthocyanins can be formed. For example in strawberries the main anthocyanins are Cyanindin-3-glucoside and Pelargonidin-3-glucoside, two relatively simple structures. In grapes on the other hand Cyanidin-, Pelargonidin-, Delphinidin-, Petunidin- and Malvidin-glucosides and di-glucosides as well as non glucosylated anthocyanidins can be found. In addition, diversity is further increased by the chemical combination of sugars with organic acids (such as acetic, succinic, caffeic acid and many more) to produce acylated anthocyanins. An example is Cyanidin-3-(acetylglucoside) in oranges. Occurrence and functionAnthocyanins occur in nearly all plant families and thus in many edible plants. In food, the main sources of anthocyanins are berries, such as blackberries, grapes, blueberries etc, and some vegetables, such as egg-plants (aubergine) and avocado. Other sources include oranges, elderberry, olives, red onion, fig, sweet potato, mango and purple corn. The natural production of anthocyanins in nature is estimated to be 109 tonnes/year ! Table 2 : Anthocyanin content in some edible plants
Anthocyanins have many different functions for the plant. They are antioxidants, protect the plant against UV-light, are a defense mechanism and are of course very important in the pollination and reproduction. The colour of many flowers is due to anthocyanins, and thus are important in attracting insects. Bee-pollinated species, such as plants from the Primulaceae are more abundant in delphinidin-anthocyanins, whereas other anthocyanins are more preferred by bumblebees or hummingbirds. Cyaninidin-3-glucoside, which is a rather common anthocyanin, has been proven to protect plants against some larvae. Other anthocyanins may have similar activities. As anti-oxidants anthocyanins protect plants from free radicals (produced by sunlicht or destruction of the plant), which may destroy the DNA, and cause cell death. UseAnthocyanins are water soluble strong colours and have been used to colour food since historical times. Extracts of berries have been used to colour drinks, pastries and other foods. There are, however, some drawbacks in the use of anthocyanins in food. Anthocyanins are water soluble, which restricts the use, and are pH dependent. When the acidity changes, the colour changes. For example the colour of red cabbage is enhanced with the addition of vinegar or other acid. On the other hand, when cooked in aluminium pans, which cause a more alkaline environment, the colour changes to purple and blue. The colour is also susceptible towards temperature, oxygen, UV-light and different co-factors. Temperature may destroy the flavylium ion, and thus causes loss of colour. Temperature also causes Maillard reactions, in which the sugar residues in the anthocyanins may be involved. Light may have a similar effect. Oxygen may destroy the anthocyanins, as do other oxidizing reagents, such as peroxides and vitamin C. Many other components in plants and foods may interact with the anthocyanins and either destroy, change or increase the colour. Quinones in apples, for example, enhance the degradation of anthocyanins, whereas the addition of sugar to strawberries stabilises the colour. All these factors limit the use of anthocyanins in foods. Some loss of colour during storage may be prevented by storing at low temperatures, in dark containers or under oxygen-free packaging. In practice the pure colours are very hard to obtain and most often (crude) extracts are used as food colours. Grape peel (E163(i)), and black currant extract (E163(iii)) are the most widely used anthocyanin mixtures in foods. Activity and toxicityAnthocyanins, when used as food colours, are not toxic and do not exceed the levels that may be ingested by consuming berries or other anthocyanin-coloured fruits. The toxicity of the highly concentrated anthocyanin mixtures that are presently sold as food supplements is not well investigated. There are indications that anthocyanins as antioxidants have health promoting effects, which include reduced risk of coronary heart disease, improved visual activity and antiviral activity. However, many of these claims are not proven scientifically. Anthocyanins are important antioxidants, but their effect on human health is still poorly understood.
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Food-Info.net is an initiative of Stichting Food-Info, The Netherlands | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||