Food-Info.net> Questions and Answers > Food Safety > Toxins
What is dioxin and how does it get in our food?
There is not such a thing as one dioxin but it is a general name for a group of compounds containing two chlorinated benzene rings, that are bonded to each other by two oxygen bridges. This last bonding is responsible for the name (di = 2, oxo = oxygen binding). The dioxin that has been of much concern in the news lately is the so called 2,3,7,8-tetrachlorine dibenzo-p-dioxin (TCDD). It is a flat molecule (see spatial picture) ; the two benzene rings are coloured grey. 4 chlorine atoms (green) are symmetrically attached to the benzene rings, and the oxygen atoms are shown in red.
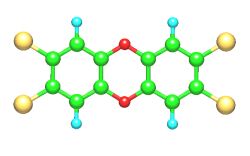
The number of chlorine atoms can differ, which results in the existence of a large group of different dioxins.
Dioxins are formed by heating chlorinated substances under poor oxygen conditions. Most dioxins are formed by the combustion of organic materials in the presence of chlorine (e.g. from table salt), where there are poor oxygen conditions. This led to the dioxin crisis a couple years ago, caused by the grazing of cows next to combustion ovens. Dioxin will not be formed in environments with enough available oxygen and the correct burning temperature.
Dioxins can also naturally be formed by white rot fungi. These are moulds growing on dead wood that form white colonies. Here dioxins are created from lignin. Dioxins can also be formed with the burning of wood, for example in a home fire or forest fire when there is poor ventilation.
Dioxins can enter the environment in different ways. They are slowly destroyed by sunlight and also through the activity of moulds in the ground. Dioxins are therefore long present in the environment.
Dioxins are not soluble in water, but very soluble in fat. Via ash particles dioxins can be found on plants, for example grass. When a cow eats the grass dioxin can be taken up by the cow and stored in its fat. Through its milk the cow can excrete the dioxin. The same happens with humans. Dioxins enter the body through food and are stored in the body fat where the dioxin can stay for a long time. Just as with cows mother milk is the most important way to excrete dioxin. Because of this human milk contains small amounts of dioxin.
Almost all animal fat contains small amounts of dioxin. In principle plant fats do not contain any dioxin. Normally the amounts of dioxin are below the maximum acceptable intake.
The daily intake of dioxin in North America. European figures are not easily available, but will not differ much.
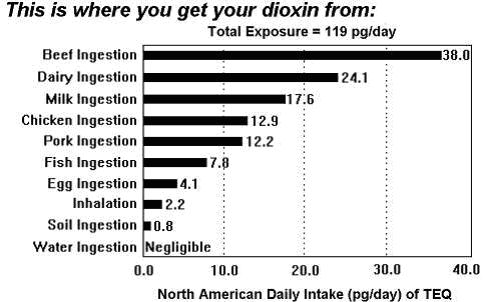
This graph illustrates the value in picograms per day as TEQ, a combination of all dioxins (toxin-equivalents).
Dioxin can only occur in relatively high amounts in food through industrial accidents (Seveso) or through chemical spoilage, as reflected by the incident in Belgium. Because it is difficult to break down it can spread in several products.
Dioxin is seen as very toxic. It is one of the most toxic (mainly) manmade substances. There exist also a few natural substances that are much more toxic, see table :
LD 50 oral for rat/mouse/guinea-pig in micrograms(=1/1000mg) per kg body weight.
Botulin |
0.00003 |
Tetanus-toxin |
0.0001 |
Difterie-toxin |
0.3 |
Paly toxin |
0.45 |
2.3.7.8-TCDD |
1 (dioxin discussed here) |
Tetrodoxine |
15 (found in fugu') |
Strychin |
500 |
Aflatoxin |
600 (found in rotten peanuts) |
Nicotin |
1000 |
Parathion |
3000 |
BPC's |
3000-30000 (?) |
Cyanide |
10000 |
DDT |
113000 |
Mercury |
120000 |
Salt |
4000000 |
The table illustrates the levels of intake of a substance per kilogram body weight at which half of the animals died. (LD 50 = lethal dose for 50% of the animals.)
Botulin (toxic substance responsible for botulism) is the most toxic substance and has a LD50 of 0.00003 micrograms per kilogram body weight. For a human of 70 kilos it is 0.0000021 mg! This calculated for dioxin is 0.07 mg and for cyanide 70 mg.
Although dioxin is very toxic, the toxicity of dioxin is in practice very hard to define: Dioxin is a molecule which can interfere with several parts of the metabolic processes of the human body. At present there is hardly any disease found which could be directly connected with dioxin.
Only chlorine acne can be said to be directly connected, caused by its chlorine bondages. Chlorine acne is quite harmless but can be very troublesome.
Dioxin appeared in several tests to be a possible mutagen and also a carcinogen, in relatively low doses, but as with almost all carcinogenic substances the causal relation is not proved. It is very rare to find the exact cause of cancer, since the process of developing cancer takes a long time. Therefore substances which cause cancer in animal tests are suspiscious, just like dioxin.
Dioxin can also cause other problems, but the fact that it is dangerous for animals used in the laboratory does not necessarily mean that it will have the same effect on humans. However, these substances are then seen as potential hazards.
The problem with dioxins is that they are only found in foods in very small amounts. This might lead to chronic exposure, which might cause problems in the (very) long term. Through this it is not possible to give the direct reason of cause relation. Therefore substances of this kind are defined as potential hazards. Necessary precautions must be taken to avoid any uptake.Only if dioxin comes free in great amounts (Seveso 1980) can direct effects be determined, which would probably be chlorine acne, and sheep death resulting from liver damage.
Dioxin is a potential dangerous substance, but the effect of intake cannot be predicted. In practice it can differ from no effect to cancer.




