Food-Info.net> Topics > Food components > Protein
Protein Structure
The properties of a protein are determined by its amino acid sequence, otherwise known as its primary structure . Depending on the nature and arrangement of the amino acids present, different parts of the molecule form secondary structure such as the alpha helices (coils') or beta sheets (flat) shown below. Further folding and reorganisation within the molecule results in higher order, or tertiary structure . Each protein consists of alpha helices, beta sheets and random parts.
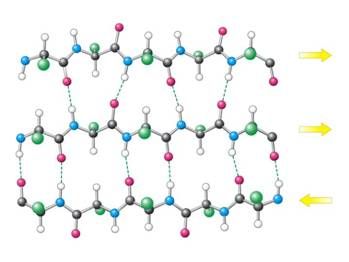
Fig. 1 : Example of a beta-sheet (arrows indicate the direction of the amino acid chain)
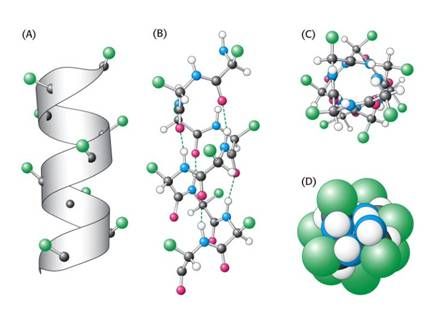
Fig. 2 : Example of an alpha-helix. A: schematic, B: molecular, C: from top, D: space filling model.
Secondary and tertiary structure represents the most stable conformation (or shape) for the molecule, and results from non-covalent interactions (ionic bonds, hydrogen bonds, hydrophobic interactions) between the various amino acid side-chains within the molecule and with the (water) molecules surrounding it. Different regions of the protein, often with distinct functions, may form structurally distinct domains. Structurally related domains are found in different proteins which perform similar functions.
The exposed surface of the protein may also be involved in interactions with other molecules, including proteins. Protein-protein interactions, for example between sub-units of enzymes, or polymeric structural proteins, results in the highest level of organisation, the quaternary structure.
Denaturing
The function of a protein (except when it is serving as food) is absolutely dependent on its three-dimensional structure. A number of agents can disrupt this structure thus denaturing the protein.
- changes in pH (acidity)
- changes in salt concentration
- changes in temperature (cooking)
- presence of reducing agents (such as bleach)
None of these agents breaks peptide bonds, so the primary structure of a protein generally remains intact when it is denatured. When a protein is denatured, it loses its function.
Often when a protein has been gently denatured and then is returned to normal physiological conditions of temperature, pH, salt concentration, etc., it spontaneously regains its function (e.g. enzymatic activity).
In prepared, cooked or canned foods, most of the proteins are denatured. Enzymes are generally totally destroyed by cooking the food.
Sources :




