Food-Info.net> Topics > Food components > Protein
Milk proteins
Milk contains hundreds of types of protein, most of them in very small amounts. The proteins can be classified in various ways according to their chemical or physical properties and their biological functions. Traditionally milk proteins were classified into caseins, whey proteins and minor proteins. Proteins present at the surface of fat globules and enzymes belong to the group of minor proteins.

Whey protein is a term often used as a synonym for milk-serum proteins, but it should be reserved for the proteins in whey from the cheese-making process. In addition to milk-serum proteins, whey protein also contains fragments of casein molecules that are split-off during renneting of the cheese milk. Some of the milk-serum proteins are also present in lower concentrations than in the original milk. This is due to heat denaturation during pasteurisation of the milk prior to cheese-making.
The three main groups of proteins in milk are distinguished by their widely different behaviour and form of existence. The caseins are easily precipitated from milk mainly by renneting or at low pH, while the serum proteins usually remain in solution. The globular serum proteins denature at moderate heating while caseins remain stable. The fat-globule membrane proteins (adhere, as the name implies, to the surface of the fat globules and are only released by mechanical action, e.g. by churning cream into butter.

Casein
Casein is a group name for the dominant class of proteins in milk. Caseins are present in all animal milk, including human milk. In cow's milk nearly 80% of the proteins are caseins, or about 26 g/litre of milk.
Caseins are divided in four sub-groups
α σ-,
αs2-, ß- and κ-casein. All four are very heterogeneous and consist each of 2-8 different genetic variants. These variants differ from each other only by a few amino acids. a- and ß- caseins have in common that amino acids are esterified to phosphoric acid. This phosphoric acid binds calcium (which are abundant in milk) to form bonds between and within molecules.
This makes that caseins easily form polymers containing several identical or different types of caseins. Due to the abundance phosphate groups and hydrophobic sites in the casein molecule, the molecular polymers formed by the caseins are very special and stable. The polymers are built up of hundreds and thousands of individual molecules and form a colloidal solution, which is what gives milk its white colour. These molecular complexes are known as casein micelles. Casein micelles, shown in figure 1, consist of a complex of sub-micelles, of a diameter of 10 to 15 nm (1 nanometer = 10-9 m). A medium-sized micelle consists of about 400 to 500 sub-micelles and may be as large as 0.4 microns (0.0004 mm).
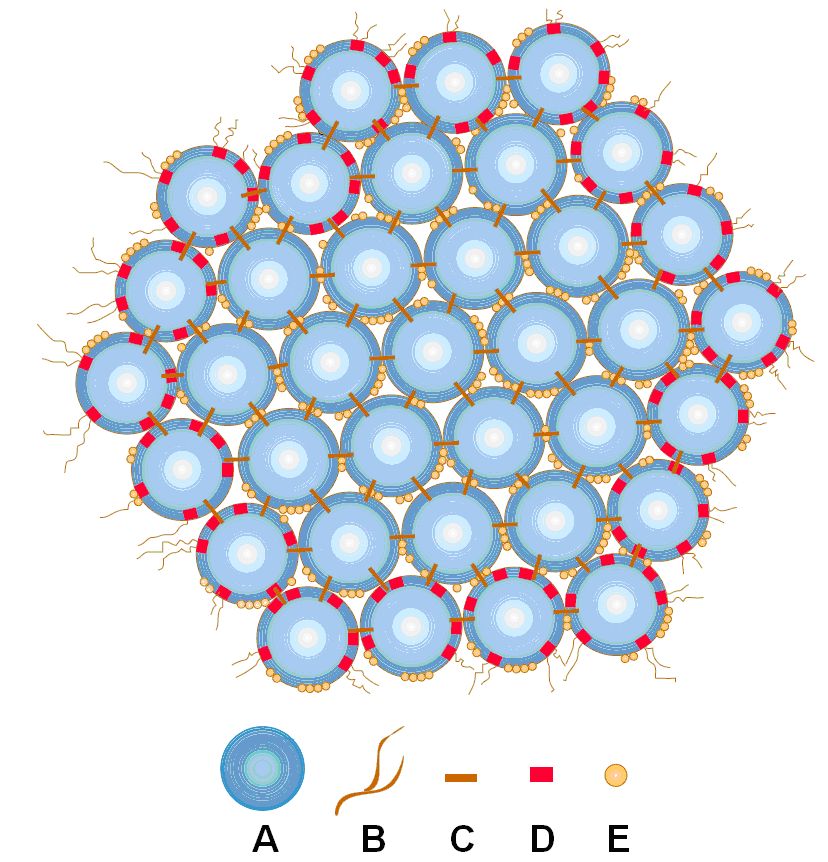
Figure 1 : A casein micelle; A: a submicelle; B: protruding chain; C: Calcium phosphate; D: κ-casein; E: phosphate groups
The calcium phosphate and hydrophobic interactions between sub-micelles are responsible for the stability of the casein micelles. The hydrophilic parts of κ-casein contain carbohydrate groups, which project from the outsides of the complex micelles (B in figure 1), giving them a “hairy” look, but more important, they stabilise the micelles against aggregation
The importance of κ-casein and its carbohydrate groups becomes evident in cheese making. The rennet, used in the first part of the cheese making process, splits the carbohydrate of the κ-casein on the surfaces of the micelles. Thereby the micelles will lose their solubility and start to aggregate to form a curd.
At low temperature the structure of the micelle is weakened, as the κ-casein chains start to dissociate and the calcium hydroxyphosphate leaves the micelle structure. The explanation of this phenomenon is that ß-casein is the most hydrophobic casein and that the hydrophobic interactions are weakened when the temperature is lowered. Hydrolysis of ß-casein to γ-casein and proteose-peptones (breakdown products) means lower yield at cheese production because the proteose-peptone fractions are lost in the whey
Other proteolytic enzymes may also used to make a curd, but often these are non-specific. Cheese made without rennet, but with vegetable proteases (for vegetarian cheese), thus often has a different taste and cheese yield will be lower.
Precipitation by acid
The pH will drop if acid is added to milk or if acid-producing bacteria are allowed to grow in milk. When milk, which has a normal pH of around 6.5-6.7, is acidified several process will occur :
- Firstly calcium phosphate, present in the casein micelle, will dissolve and form ionised calcium, which will penetrate the micelle structure and create strong internal calcium bonds.
- Secondly the pH of the solution will approach the iso-electric points of the individual casein types. The iso-electric point is the point in which the solubility of the caseins is at the lowest and is in the range of pH 4.2-4.7.
Both methods of action initiate a change within the micelles, starting with growth of the micelles through aggregation and ending with a more or less dense coagulum.
These processes occur in fermented milk. In fermented milk, such as yoghurt the bacteria also produce polysaccharides which cause the creaminess of the coagulum.
The acid precipitated casein redissolves if a large excess of sodium hydroxide is added. The resulting sodium caseinate is used as a food ingredient mainly for its excellent emulsifying properties. The original micelle structure cannot be restored by adding hydroxide.

Fig 2 : Separation of curds and whey
Whey proteins
Whey protein is the name commonly applied to milk serum proteins, but technically only contains the proteins present in whey, obtained during cheese making. If the casein is removed from skimmed milk by addition of mineral acid, there remains in solution a group of proteins which are called milk serum proteins. These are very similar to ‘true' whey proteins, hence the common name.
Whey proteins form approximately 20% of the protein fraction in milk. The whey proteins are very soluble and can be separated into the following groups:
- α-lactalbumin
- ß-lactoglobulin
- Blood Serum Albumin
- Immunoglobulins
- Miscellaneous proteins and polypeptides
Whey proteins in general, and a-lactalbumin in particular, have very high nutritional values. Their amino acid composition is very close to that which is regarded as a biological optimum. Whey protein derivatives are widely used in the food industry.
Whey proteins denature during heating which causes aggregation of the whey proteins mainly with the casein micelles
Whey proteins are isolated on an industrial scale by membrane technology
α-lactalbumin
This protein may be considered to be the typical whey protein. It is present in milk from all mammals and plays a significant part in the synthesis of lactose (milk sugar).
ß-lactoglobulin
This protein is found only in ungulates (‘hoofed' animals) and is the major whey protein component of milk from cows. If milk is heated to over 60 °C, denaturation is initiated where the reactivity of the sulphur-amino acid of ß-lactoglobulin plays a prominent part. At high temperatures sulphurous compounds are gradually released. These sulphurous compounds are partly responsible for the “cooked” flavour of heat treated milk.
Immunoglobulins
The immunoglobulins play an important role in the protection of the newborn animal (or human) against bacteria and diseases.
Lactoferrin
Lactoferrin is a glycoprotein that belongs to the iron transporter or transferrin family. It was originally isolated from bovine milk, but it is also present in milk of other animals. In addition to its presence in milk, it is also found in exocrine secretions of mammals.
Lactoferrin is considered a multifunctional or multi-tasking protein. It appears to play several biological roles. Owing to its iron-binding properties, lactoferrin is thought to play a role in iron uptake by the intestinal mucosa of the suckling neonate. It also appears to have antibacterial, antiviral, antifungal, anti-inflammatory, antioxidant and immunomodulatory activities. These activities are being studied extensively.
Lactoperoxidase
Lactoperoxidase has been identified as an antimicrobial agent in milk, saliva and tears. Lactoperoxidase is a natural bacterial defence system through the oxidation of thiocyanate ions (SCN-) by hydrogen peroxide. These are both present in biological fluids and together with lactoperoxidase are termed the lactoperoxidase system (LP-s). LP-s has proven to be both bactericidal and bacteriostatic to a wide variety of micro-organisms, while having no effect on the proteins and enzymes of the organisms producing it.
Minor Proteins
Membrane proteins
Membrane proteins are a group of proteins that form a protective layer around fat globules to stabilise the emulsion of fat droplets in the milk. Some of the proteins contain lipid residues and are called lipoproteins. Membrane globulin proteins are the smallest protein fraction in milk, approximately 1.5% of the total protein fraction.
The lipids and the hydrophobic amino acids of those proteins make the molecules direct their hydrophobic sites towards the fat surface, while the less hydrophobic parts are oriented towards the water.
Enzymes in milk
Phospholipids and lipolytic enzymes in particular are adsorbed within the membrane structure.
The enzymes in milk come either from the mother animal or from bacteria. The former are normal constituents of milk. The latter, bacterial enzymes, vary in type and abundance according to the nature and size of the bacterial population and are not considered here.
Several of the enzymes in milk are utilised for quality testing and control. Among the more important ones are peroxidase, phosphatase and lipase.
Lactoperoxidase
Peroxidase transfers oxygen from hydrogen peroxide (H2O2) to other readily oxidisable substances. This enzyme is inactivated if the milk is heated to 80 °C for a few seconds, a fact which can be used to prove the presence or absence of peroxidase in milk and thereby check whether or not a pasteurisation temperature above 80 °C has been reached. This test is called Storch's peroxidase test.
Phosphatase
Phosphatase has the property of being able to split certain phosphoric-acid esters into phosphoric acid and the corresponding alcohols. The presence of phosphatase in milk can be detected by adding a phosphoric-acid ester and a reagent that changes colour when it reacts with the liberated alcohol. A change in colour reveals that the milk contains phosphatase. Phosphatase is destroyed by ordinary pasteurisation (72 °C for 15 – 20 seconds), so the phosphatase test can be used to determine whether the pasteurisation temperature has actually been attained.
Lipase
Lipase splits fat into glycerol and free fatty acids. Excess free fatty acids in milk and milk products result in a rancid taste. The action of this enzyme seems, in most cases, to be very weak, though the milk from certain animals may show strong lipase activity. The quantity of lipase in milk is believed to increase towards the end of the lactation cycle. No reactions occur between milk lipase and fat as long as the surface of the fat globules is intact, but as soon as the surface is destroyed the enzyme lipase has the opportunity to find substrate and free fatty acids are released. When milk has been pumped cold with a faulty pump, or after homogenisation of cold milk without pasteurisation free fatty acids are immediately formed. The fatty acids and some other products of this enzymatic reaction give a “rancid” flavour to the product.
Sources:






