|
|||||
|
|||||||
|
    An initiative of :Stichting Food-Info
|
| Food-Info.net> Topics > Food components > Protein Amino AcidsProteins are made of large numbers of small building blocks, the amino acids. In proteins 20 amino acids can be found, which are chemically characterized by the structure RCH(NH2)COOH. The nitrogen (N) and two hydrogen (H) atoms comprise the amino group, -NH2, and the acid entity is the carboxyl group, -COOH. The R- group is the side chain, which determines the actual amino acid. Only one amino acid, proline, has a slightly different structure, see the table below. Amino acids link to each when the carboxyl group of one molecule reacts with the amino group of another molecule, creating a peptide bond -C(=O) NH- and releasing a molecule of water (H2O). A peptide is a compound consisting of 2 or more amino acids. Oligopeptides have 10 or fewer amino acids. Polypeptides and proteins are chains of 10 or more amino acids, but peptides consisting of more than 50 amino acids are classified as proteins.
The term "essential amino acid" refers to an amino acid that is required to meet physiological needs and must be supplied in the diet. Arginine is synthesized by the body, but at a rate that is insufficient to meet growth needs. Methionine is required in large amounts to produce cysteine if the latter amino acid is not adequately supplied in the diet. Similarly, phenylalanine can be converted to tyrosine, but is required in large quantities when the diet is deficient in tyrosine. Tyrosine is essential for people with the disease phenylketonuria (PKU) whose metabolism cannot convert phenylalanine to tyrosine. Isoleucine, leucine, and valine are sometimes called "branched-chain amino acids" because their carbon chains are branched. StereochemistryIn all twenty amino acids, except glycine, the carbon atom with the amino group is attached to four different substituents. The tetrahedral bond angles of carbon and the asymmetry of the attachments make it possible for amino acids to have two non-superimposable structures, the L and R forms, which are mirror images of each other (compare left and right hand). Only L-amino acids are found in proteins. L-amino acids have the amino group to the left when the carboxyl group is the top, as illustrated below. The wedge bonds are above the display plane and the dotted bonds are below the display plane.
Formation of a peptide from two amino acids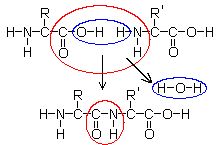 This illustration shows the reaction of two amino acids, where R and R' are any functional groups from the table above. The blue circle shows the water H2O) that is released, and the red circle shows the resulting peptide bond (-C(=O) NH-). The reverse reaction, i.e., the breakdown of peptide bonds into the component amino acids, is achieved by hydrolysis. Many commercial food products use hydrolyzed vegetable proteins as flavouring agents. Soy sauce is produced by hydrolyzing soybean and wheat protein by fungal fermentation or by boiling with acid solutions. Monosodium glutamate (MSG), a flavour enhancer, is a sodium salt of glutamic acid that is found naturally in seaweed and fermented soy products. Sources :
|
|
| ||||||||||||||||||||||||||
| Food-Info.net is an initiative of Stichting Food-Info, The Netherlands | ||||||||||||||||||||||||||||||